What Is Immunotherapy for Cancer and How Does It Work
Immunotherapy is something that comes up more and more when discussing cancer treatment, often as a newer option or something that sounds promising on paper.


Jump to:
In reality, it’s not quite that simple. It works in a very different way from traditional treatments, and understanding that difference is what tends to matter most.
What immunotherapy is actually doing
Immunotherapy is about helping the immune system recognise cancer.
The immune system is constantly scanning for things that shouldn’t be there. Bacteria, viruses, and abnormal cells. In theory, cancer should fall into that category. But it doesn’t always trigger a response.
That’s because cancer cells can send signals that effectively tell the immune system to ignore them. So the problem isn’t always that the body can’t fight the cancer. It’s that it doesn’t realise it needs to.
Immunotherapy works by interfering with those signals.
In simple terms, it removes the “brakes” that stop immune cells from acting. Once those brakes are lifted, the immune system can start to recognise and attack the cancer more effectively.
That’s the principle behind most modern immunotherapy cancer treatment approaches we use in practice, particularly the group known as checkpoint inhibitors.
When immunotherapy is used in cancer care
Immunotherapy isn’t suitable for every cancer or every patient. The decision to use it usually depends on a few key factors.
One of the most important is whether the tumour shows certain biomarkers. In lung cancer, for example, PD-L1 expression is often used to guide whether immunotherapy is likely to be effective. In patients with higher PD-L1 expression, immunotherapy is often more effective than chemotherapy alone, although it doesn’t apply in every case.
It’s also used differently depending on the stage of cancer. In advanced cases, it may be used as a primary treatment. In earlier stages, it can be used alongside surgery or radiotherapy.
And in some cancers, such as melanoma, immunotherapy has changed outcomes quite substantially over time, with long-term responses now seen in a proportion of patients. That wasn’t something we expected even 10-15 years ago.
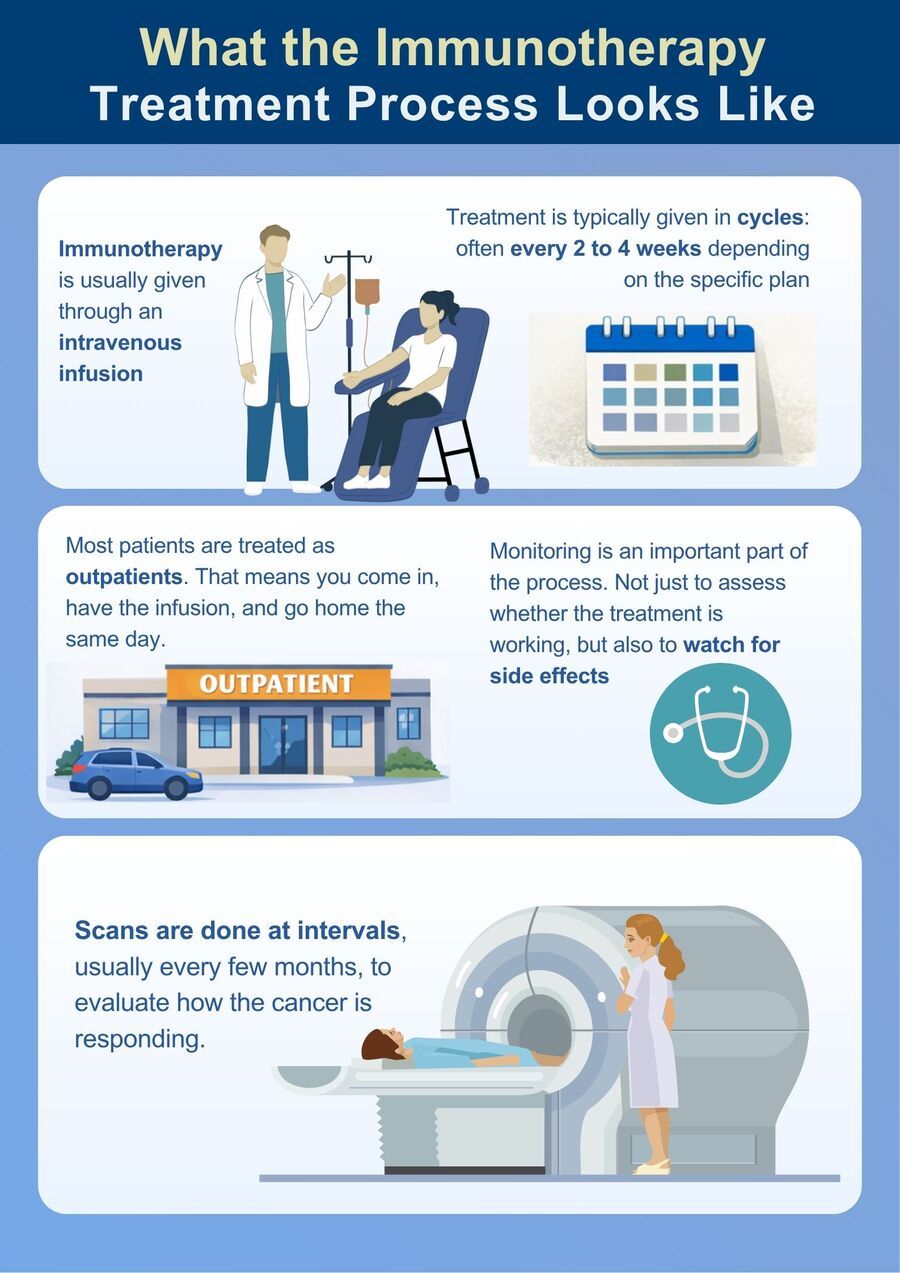
What the treatment process looks like
In practical terms, immunotherapy is usually given through an intravenous infusion.
Treatment is typically delivered in cycles, often every two to four weeks, depending on the specific drug and the plan being followed.
Most patients are treated as outpatients. That means you come in, have the infusion, and go home the same day.
Monitoring is an important part of the process. Not just to assess whether the treatment is working, but also to watch for side effects, which can behave quite differently from those seen with chemotherapy.
Scans are done at intervals, usually every few months, to evaluate how the cancer is responding.
How it differs from chemotherapy
Chemotherapy and immunotherapy are often grouped together, but they approach the problem from different angles.
Chemotherapy is more direct. It targets fast-growing cells and aims to destroy them. That includes cancer cells, but also some healthy cells, which is why side effects can happen.
Immunotherapy is less direct. It doesn’t attack the cancer itself. Instead, it changes the conditions around it, so the immune system can step in.
This difference matters when deciding between treatments or when combining them. In some cases, chemotherapy is used to reduce the tumour burden first. Immunotherapy can then help maintain a longer-term immune response.
The combination of approaches is now common, particularly in lung cancer care, where treatment plans are tailored based on both clinical findings and molecular testing.
Side effects and what to expect
There’s sometimes an assumption that immunotherapy is easier to tolerate than chemotherapy. That’s not always the case.
The side effects are different rather than milder.
Because immunotherapy activates the immune system, it can sometimes lead to inflammation in normal tissues. This might affect the lungs, skin, liver, or endocrine system.
For example, some patients develop a skin rash or itching. Some notice diarrhoea or abdominal pain if the bowel becomes inflamed. Some become unusually tired because the thyroid is affected. Less commonly, we see inflammation in the lungs, which can cause breathlessness or a new cough.
Most side effects are manageable, particularly when identified early. But they do require attention.
That’s why follow-up is structured quite carefully, even when the treatment itself feels relatively straightforward.
How decisions are made around immunotherapy
Deciding whether to use immunotherapy isn’t a single-step decision.
It involves looking at:
- the type and stage of cancer
- molecular and biomarker testing
- overall health and fitness
- previous treatments, if any
In lung cancer, these decisions are often made within a multidisciplinary team. There’s a balance between what is technically possible and what is likely to offer meaningful benefit.
A large part of this also comes down to having the right oncology specialist involved. Someone who is used to working with these treatments and can interpret how they apply in the context of an individual case, rather than in general terms.
The truth is, not every patient will benefit from immunotherapy. And in some cases, other treatments may be more appropriate.
The aim is always to match the treatment to the biology of the cancer, rather than applying a one-size-fits-all approach.
About Dr James Wilson
Dr James Wilson is a consultant oncologist specialising in lung cancer and systemic treatments such as immunotherapy. Based in Central London, he works full-time in private practice, helping patients understand their options, make clear treatment decisions, and move forward with a calm, practical approach, particularly when timing and clarity matter most.