Lung Cancer Remission Rates: Treatment Options by Stage and Type
If you've been diagnosed with lung cancer, you've done some online research, then one of your next questions is likely: "What are lung cancer remission rates?"
The answer depends on several factors: the type of lung cancer you have, the stage at diagnosis, whether your tumour carries specific genetic mutations, and which treatment options are available to you. Modern lung cancer treatment has changed dramatically in recent years. Targeted therapies, immunotherapy, and advanced radiation therapy techniques like SABR are achieving lung cancer remission rates that would have seemed impossible a decade ago.
This page explains lung cancer remission rates by stage and type, how genetic mutations influence your prognosis, and which treatment approaches offer the best chance of long-term control. Whether you're a lung cancer patient researching options for yourself, or someone seeking cancer information for a person you care about, you'll find the clinical data and treatment pathways that matter most.


Jump to:
- Get Your Personal Treatment Plan
- What Are Lung Cancer Remission Rates?
- Remission Rates by Lung Cancer Type
- Non-Small Cell Lung Cancer (NSCLC) Remission Rates
- Understand Your Lung Cancer Stage & Options
- Small Cell Lung Cancer (SCLC) Remission Rates
- How Genetic Mutations Impact Lung Cancer Remission
- EGFR-Positive Lung Cancer Remission
- ALK-Positive Lung Cancer Remission
- Other Targetable Mutations (ROS1, KRAS G12C, BRAF)
- Perioperative Immunotherapy for Resectable NSCLC
- Access Advanced Treatment Quickly
- Stereotactic Radiotherapy (SABR) for Early-Stage Lung Cancer
- Targeted Therapy for Genetic Mutations
- Immunotherapy for Lung Cancer
- Proton Beam Therapy for Lung Cancer
- Surgery and Adjuvant Therapy
- Treatment for Oligometastatic Lung Cancer
- Explore Oligometastatic Treatment
- How Long Does Lung Cancer Remission Last?
- Factors That Influence Remission Duration
- Median Remission Duration by Treatment Type
- What Happens When Lung Cancer Remission Ends?
- Second-Line Treatment Options
- Re-Treatment with SABR for New Lung Nodules
- Clinical Trials for Resistant Lung Cancer
- Palliative Care to Maintain Quality of Life
- Monitoring Your Lung Cancer for Recurrence
- Standard Monitoring: CT Scans and Clinical Exams
- Advanced Blood Monitoring: Circulating Tumour DNA (ctDNA)
- Get Comprehensive Surveillance
- Symptoms That Require Urgent Review
- Living Well During Lung Cancer Remission
- Managing Physical Side Effects After Treatment
- Emotional Challenges of Remission
- Lifestyle Modifications to Support Remission
- Does Smoking History Affect Lung Cancer Remission Rates?
- Frequently Asked Questions About Lung Cancer Remission
- What is the difference between remission and cure in lung cancer?
- Can stage 4 lung cancer go into remission?
- What are the early signs of lung cancer remission?
- Is lung cancer remission possible without chemotherapy?
- How often will I need scans during lung cancer remission?
- Can lung cancer come back after 5 years of remission?
- Take Control of Your Treatment Today
- Dr James Wilson's Approach to Lung Cancer Remission
- Rapid Access to Advanced Lung Cancer Treatment
- SABR Expertise: 90% Control Rate in Early-Stage Lung Cancer
- Coordinated Care Across London's Leading Cancer Hospitals
- Personalised Treatment Based on Your Cancer's Genetic Profile
- Take the Next Step Toward Lung Cancer Remission
- Start Your Journey to Remission
Get Your Personal Treatment Plan
Discuss your diagnosis, staging, and personalised remission strategy with Dr James Wilson. Same-day appointments available for urgent cases.
Book Your Consultation – Available Within 48 Hours
What Are Lung Cancer Remission Rates?
Remission means your cancer has responded to treatment and is no longer detectable on scans, or has shrunk significantly and remains stable. It doesn't always mean cure – some lung cancer survivors remain in remission for years or decades, while others eventually experience cancer recurrence that requires further treatment.
Oncologists typically describe remission in three ways:
Complete remission (complete response): No evidence of cancer on CT, PET, or other imaging. Blood tumour markers, if previously elevated, have returned to normal. All cancer cells appear to have been eliminated, though microscopic disease may still exist.
Partial remission (partial response): The tumour has shrunk by at least 30% and remains stable. Symptoms have improved. Further treatment may be needed, but disease is controlled. Tumour cells remain visible on imaging but are responding to therapy.
Stable disease: The cancer hasn't grown or spread, even if it hasn't shrunk significantly. This is increasingly recognised as a valuable outcome in stage 4 lung cancer, particularly with immunotherapy or targeted therapies that can hold cancer in check for extended periods.
Understanding lung cancer remission rates requires looking at survival statistics. According to cancer statistics from Cancer Research UK, the American Cancer Society, and the National Cancer Institute, lung cancer survival rates have improved substantially over the past decade, particularly for patients with targetable mutations or immunotherapy-sensitive disease.
The rates below reflect complete remission and partial remission combined, as both represent meaningful lung cancer treatment success:
| Lung Cancer Type | Stage | 5-Year Survival Rate | Primary Treatment |
|---|---|---|---|
| NSCLC | Stage 1A | 77-92% | Surgery or SABR |
| NSCLC | Stage 1B | 68-83% | Surgery or SABR |
| NSCLC | Stage 2A | 60-75% | Perioperative immunotherapy + surgery or radiotherapy |
| NSCLC | Stage 2B | 53-67% | Perioperative immunotherapy + surgery ot radiotherapy |
| NSCLC | Stage 3A | 36-50% | Perioperative immunotherapy + surgery, or concurrent chemoradiotherapy |
| NSCLC | Stage 3B-C | 26-36% | Concurrent chemoradiotherapy + immunotherapy/targeted therapy |
| NSCLC | Stage 4 (driver mutation) | 15-50% | Targeted therapy ± local therapy |
| NSCLC | Stage 4 (immunotherapy-sensitive) | 15-30% | Immunotherapy + chemotherapy |
| Small cell lung cancer | Limited stage | 20-40% | Chemotherapy + radiation therapy |
| Small cell lung cancer | Extensive stage | 2-5% | Chemotherapy ± immunotherapy |
These lung cancer survival rate figures represent general population data. Your individual lung cancer remission rate may be significantly better depending on factors like genetic mutations, treatment access, and overall health. Remember that your doctor can only talk to you about population-based averages. My job, as your oncologist, is to come up with a strategy that gets the best chance of success, however you define it, for you.
Remission Rates by Lung Cancer Type
Non-Small Cell Lung Cancer (NSCLC) Remission Rates
NSCLC accounts for 85% of lung cancer cases in the UK. It includes lung adenocarcinoma (most common in never-smokers and women), squamous cell carcinoma (more common in smokers), and large cell carcinoma.
Stage 1 lung cancer outcomes
Early-stage NSCLC offers the highest lung cancer remission rates because the cancer is localised and hasn't spread to lymph nodes or distant organs.
Surgical resection (lobectomy or segmentectomy) achieves complete remission in 70-85% of stage 1 patients at five years. These lung cancer survivors are genuinely cured – they die of other causes, not cancer recurrence. According to local cancer statistics and cancer stat facts, stage 1 lung cancer treated with surgery has among the best survival statistics of any cancer type when detected early through lung cancer screening programmes.
For lung cancer patients who cannot have lung cancer surgery due to poor lung function (often related to chronic obstructive pulmonary disease), heart disease, or other medical conditions, stereotactic ablative radiation therapy (SABR) achieves similar outcomes. In my own practice using CyberKnife SABR, we see 90% local control at five years for stage 1 NSCLC – meaning the treated tumour does not grow back. That's functionally equivalent to surgical cure for most patients.
Stage 2 NSCLC involves slightly larger tumours or limited lymph node involvement. The treatment landscape has been transformed by perioperative immunotherapy – giving immunotherapy before and after surgery rather than chemotherapy alone. This represents one of the most significant advances in clinical oncology for lung cancer in the past decade.
Stage 3 NSCLC outcomes
Stage 3 NSCLC is locally advanced – the cancer has spread to multiple lymph nodes in the chest but hasn't metastasised to other organs. Treatment choices are more complex.
Some stage 3A lung cancer patients are candidates for perioperative immunotherapy followed by surgery and then completion immunotherapy. Based on the CheckMate-77T clinical trial, this approach achieves event-free survival of 74% at three years compared to 63% with chemotherapy alone – a genuinely transformative advance for resectable stage 3A disease.
For stage 3A patients whose tumours are borderline resectable, neoadjuvant immunotherapy plus chemotherapy (CheckMate-816) achieves pathological complete response – meaning no viable cancer cells found at surgery – in 24% of patients. These patients have exceptionally low cancer recurrence rates.
Others receive concurrent chemoradiotherapy (chemotherapy and radiation therapy given together), followed by a year of immunotherapy consolidation with durvalumab. This approach, based on the PACIFIC clinical trial, has transformed stage 3 NSCLC outcomes and improved overall survival substantially. Five-year survival has increased from 36% to nearly 43% with the addition of immunotherapy.
Stage 3B and 3C disease is generally not operable. Concurrent chemoradiotherapy followed by immunotherapy achieves durable remission in 26-36% of lung cancer patients at five years.
Oligometastatic NSCLC
Oligometastatic disease sits between stage 3 and typical stage 4 disease. You have a small number of metastases (typically 1-5 sites) that can all be treated with local therapy like SABR, while systemic therapy controls any microscopic disease.
This is an area where treatment philosophy has shifted significantly in lung cancer research. We now know that aggressive local treatment combined with modern systemic therapy can achieve long-term lung cancer remission in carefully selected patients.
In the SABR-COMET trial, patients with oligometastatic cancer (including lung) who received SABR to all visible metastases had a median survival of 50 months compared to 28 months with systemic therapy alone. Some of these cancer survivors remain in remission five years later – lung cancer survival outcomes that looked like stage 2 or 3 disease rather than stage 4.
If you have oligometastatic NSCLC, particularly with a targetable mutation or immunotherapy-sensitive disease, this is worth discussing in detail. The difference between "stage 4 cancer managed with ongoing treatment" and "oligometastatic disease treated aggressively for potential long-term remission" is profound.
Understand Your Lung Cancer Stage & Options
Get clarity on your specific stage, genetic profile, and which treatments offer you the best remission rates. Consultations available same or next day.
Arrange Your Consultation TodaySmall Cell Lung Cancer (SCLC) Remission Rates
Small cell lung cancer is more aggressive than NSCLC. It grows quickly, spreads early, and is more commonly caused by smoking. According to cancer incidence data, it accounts for approximately 15% of lung cancer cases in the UK. Small cell lung cancer is a leading cause of cancer death in the UK, with lung cancer deaths from SCLC representing a disproportionate share given its lower incidence compared to NSCLC.
Limited stage small cell lung cancer
Limited stage means the cancer is confined to one side of the chest and can be encompassed within a single radiation therapy field.
Cancer treatment is concurrent chemotherapy (usually platinum + etoposide) and radiation therapy, followed by prophylactic cranial irradiation (PCI) to reduce the risk of brain metastases.
Complete remission is achieved in 70-80% of small cell lung cancer patients initially. However, small cell lung cancer frequently relapses. Five-year survival is 20-40%, meaning long-term lung cancer remission is possible but less common than in NSCLC. The median survival for limited-stage small cell lung cancer is approximately 15-20 months, though long-term survivors do exist.
Extensive stage small cell lung cancer
Extensive stage small cell lung cancer has spread beyond one side of the chest – to the other lung, pleura, distant lymph nodes, liver, bones, or brain. This is considered metastatic lung cancer.
Chemotherapy achieves partial remission or complete remission in 60-70% of patients initially. However, remission duration is typically short – measured in months rather than years. Five-year survival is only 2-5%, with median survival around 8-13 months.
The addition of immunotherapy (atezolizumab or durvalumab) to first-line chemotherapy has extended survival modestly, from a median of 10 months to 12-13 months. Durable remissions beyond two years remain rare in extensive stage small cell lung cancer, but they do occur – particularly in lung cancer patients whose cancer responds well to initial treatment.
Spontaneous remission – cancer disappearing without treatment – is extraordinarily rare in small cell lung cancer and should not be relied upon. Active cancer treatment is essential.

How Genetic Mutations Impact Lung Cancer Remission
One of the most important developments in lung cancer treatment over the past 15 years has been the discovery that certain genetic mutations make tumours vulnerable to targeted therapies.
If your lung cancer carries one of these mutations, your lung cancer remission rates may be significantly better than the general population figures above – sometimes dramatically so, with overall survival extending years beyond what historical cancer statistics would predict.
This is why biomarker testing is essential for anyone diagnosed with NSCLC, particularly lung adenocarcinoma. According to guidance from the National Comprehensive Cancer Network, the American Lung Association, and major lung cancer research organisations, comprehensive molecular testing should be performed on all advanced lung cancer cases.
EGFR-Positive Lung Cancer Remission
EGFR (epidermal growth factor receptor) mutations occur in approximately 10-15% of lung cancer cases in the UK, and up to 40% in East Asian populations. They're more common in never-smokers, women, and lung cancer patients with adenocarcinoma.
First-line cancer treatment is an EGFR tyrosine kinase inhibitor (TKI) – usually osimertinib (Tagrisso), which is now standard in the UK for both early exon 19 deletions and exon 21 L858R mutations.
Lung cancer remission rates are exceptional. In the FLAURA trial, 80% of patients with metastatic EGFR-positive NSCLC achieved objective response (complete remission or partial remission) on osimertinib. Median duration of response was 17.2 months, with many lung cancer patients remaining in remission for two to three years or longer. The overall survival for EGFR-positive metastatic lung cancer has improved from a median of 12-15 months with chemotherapy to over 38 months with osimertinib.
For patients with early-stage EGFR-positive NSCLC who have been treated with lung cancer surgery, adjuvant osimertinib reduces the risk of cancer recurrence by 80%. The ADAURA trial showed disease-free survival of 89% at three years in stage 1B-3A patients who received three years of osimertinib after surgery, compared to 52% in the placebo group.
This is genuinely transformative. Lung cancer patients who would historically have had a 50-50 chance of recurrence now have nearly a 90% chance of remaining in remission.
The NorthStar trial: Combining osimertinib with local consolidative therapy
One of the most significant developments presented at ESMO Congress 2025 was the NorthStar trial, which demonstrated that adding local consolidative therapy (LCT) – surgery, radiotherapy (including stereotactic radiotherapy), or both – to osimertinib substantially improves outcomes in metastatic EGFR-positive NSCLC.
In this randomised phase II trial, lung cancer patients with EGFR exon 19 deletion or L858R mutations who achieved disease control after starting osimertinib were randomised to either continue osimertinib alone or receive osimertinib plus LCT to all sites of disease.
The results were striking: median progression-free survival was 25.3 months with osimertinib plus LCT compared to 17.5 months with osimertinib alone – a 34% reduction in the risk of progression (hazard ratio 0.66, p=0.025).
This benefit was seen across multiple subgroups:
-
Patients with EGFR exon 19 deletion: 39.8 months versus 22.8 months
-
Patients with EGFR L858R mutation: 19.1 months versus 11.0 months
-
Even patients with more than three metastases: 20.7 months versus 15.9 months
Patients who received comprehensive LCT (treating all visible disease sites) did particularly well, with a median PFS of 33.1 months – nearly three years of remission on first-line treatment.
This represents a paradigm shift in how we approach metastatic EGFR-positive lung cancer. Rather than viewing metastatic disease as uniformly incurable, the NorthStar data suggest that aggressive local treatment of all visible metastases – using SABR, surgery, or both – combined with ongoing targeted therapy can achieve remission durations approaching those seen in early-stage disease.
The treatment was well tolerated, with only three cases of grade 3 toxicity related to LCT (one pneumonitis, one arterial injury, one empyema).
What does this mean practically? If you have metastatic EGFR-positive NSCLC with a limited number of disease sites (typically 1-5 metastases), and your cancer responds well to osimertinib during the first 3-4 months of treatment, adding local therapy to all remaining visible disease may extend your remission from 17-18 months to 25-40 months.
This is precisely the oligometastatic treatment paradigm I've been offering patients for several years, and the NorthStar trial now provides randomised evidence that this approach works specifically for EGFR-positive disease.
When EGFR-targeted therapy stops working, resistance develops through various mechanisms – most commonly the T790M mutation (which osimertinib was designed to overcome) or other bypass pathways. Second-line and third-line treatment options exist, including newer-generation TKIs, chemotherapy, and increasingly, combination approaches with immunotherapy or chemotherapy.
ALK-Positive Lung Cancer Remission
ALK (anaplastic lymphoma kinase) rearrangements occur in 3-5% of NSCLC cases, typically in younger patients, never-smokers, and those with lung adenocarcinoma.
ALK-positive lung cancer responds exceptionally well to ALK inhibitors. We now have four generations of drugs:
-
First generation: Crizotinib (good initial response but limited brain penetration)
-
Second generation: Alectinib, brigatinib, ceritinib (better brain penetration, longer remission)
-
Third generation: Lorlatinib (effective against most resistance mutations, excellent CNS activity)
In the ALEX trial, alectinib achieved an objective response rate of 83% in previously untreated metastatic ALK-positive NSCLC. Median progression-free survival was 34.8 months – nearly three years of lung cancer remission on a single oral therapy.
Perhaps more remarkably, some lung cancer survivors remain in remission for five years or longer on ALK inhibitors. The five-year overall survival for metastatic ALK-positive NSCLC is now approaching 60-65% with sequential ALK inhibitor therapy – outcomes that make stage 4 lung cancer look more like a chronic illness than a terminal diagnosis. This represents a complete transformation in lung cancer survival statistics for this molecular subtype.
For lung cancer patients with early-stage ALK-positive NSCLC treated with surgery, adjuvant alectinib is now being investigated in clinical trials and is likely to become standard, mirroring the success of adjuvant osimertinib in EGFR-positive disease.
Other Targetable Mutations (ROS1, KRAS G12C, BRAF)
ROS1 rearrangements (1-2% of NSCLC) respond to crizotinib and entrectinib, with similar lung cancer remission rates to ALK-positive disease – 70-80% objective response rates and median progression-free survival of 15-20 months.
KRAS G12C mutations (13% of NSCLC in Western populations) are now targetable with sotorasib and adagrasib. These drugs achieve objective response rates of 35-40% and median progression-free survival of 6-12 months. While not as dramatic as EGFR or ALK inhibitors, this represents a meaningful advance for a mutation that was previously considered "undruggable."
BRAF V600E mutations (2% of NSCLC) respond to dabrafenib plus trametinib (the same combination used in breast cancer-related BRAF mutations, bladder cancer, ovarian cancer, and melanoma), achieving objective response rates of 60-65% and median progression-free survival of 10-11 months.
MET exon 14 skipping mutations (3-4% of NSCLC) respond to capmatinib or tepotinib, with objective response rates of 40-50%.
RET rearrangements (1-2% of NSCLC) respond to selpercatinib or pralsetinib, achieving response rates of 60-70%.
NTRK fusions (rare, <1% of NSCLC) respond to larotrectinib or entrectinib with response rates of 70-80% and remarkably durable remissions – some lung cancer patients remain progression-free for five years or longer.
The key message: if your lung cancer carries any of these mutations, targeted therapy should be first-line treatment. Response rates are generally superior to chemotherapy, side effects are more manageable, and quality of life during remission is considerably better. Across all these molecular subtypes, the lung cancer remission rate is substantially higher than with chemotherapy alone.
PD-L1 Expression and Immunotherapy Response
PD-L1 (programmed death-ligand 1) is a protein expressed on some lung cancer cells that helps them evade the immune system. Tumours with high PD-L1 expression (≥50% of tumour cells) respond better to immunotherapy.
Pembrolizumab (Keytruda) as monotherapy achieves objective response rates of 40-45% in previously untreated metastatic NSCLC with PD-L1 ≥50%. More importantly, responses tend to be durable – the five-year survival rate in KEYNOTE-024 was 32% for pembrolizumab compared to 16% for chemotherapy.
This durability is immunotherapy's hallmark. While response rates may be lower than with targeted therapy, lung cancer patients who do respond often remain in remission for years, sometimes even after stopping treatment. Some oncologists now use the term "functional cure" for cancer survivors who remain disease-free five years after completing immunotherapy - though that is not a term I use.
For patients with PD-L1 expression 1-49%, or even PD-L1 negative disease, combination immunotherapy plus chemotherapy achieves higher response rates (45-50%) than chemotherapy alone, and extends median survival by 4-6 months.
Tumour mutational burden (TMB) – the number of mutations in cancer DNA – also predicts immunotherapy response. Lung cancer patients with high TMB (≥10 mutations per megabase) achieve objective response rates of 45-50% with pembrolizumab, regardless of PD-L1 status.
This is why comprehensive biomarker testing matters. PD-L1, TMB, and driver mutations should all be assessed at diagnosis to guide first-line treatment decisions and optimise lung cancer remission rates.
Treatment Approaches That Achieve Lung Cancer Remission
Perioperative Immunotherapy for Resectable NSCLC
One of the most significant advances in translational lung cancer research over the past five years has been the recognition that giving immunotherapy before and after lung cancer surgery – perioperative treatment – dramatically improves outcomes compared to surgery and chemotherapy alone.
The CheckMate-77T trial compared perioperative nivolumab (immunotherapy given before surgery, then continued for one year afterward) plus chemotherapy versus chemotherapy alone in stage 2-3A resectable NSCLC.
The results were striking: event-free survival at three years was 74% with perioperative nivolumab compared to 63% with chemotherapy alone. This represents an 11% absolute reduction in cancer recurrence risk – one of the largest improvements seen in any recent lung cancer treatment clinical trial. Overall survival data is still maturing but early indications suggest substantial benefit.
The CheckMate-816 trial established that neoadjuvant immunotherapy – giving nivolumab plus chemotherapy before surgery – achieves pathological complete response in 24% of lung cancer patients. This means that when the surgeon removes the tumour, pathology shows no viable cancer cells remaining – only scar tissue. These patients have exceptionally low cancer recurrence rates and improved lung cancer survival.
For comparison, neoadjuvant chemotherapy alone achieves pathological complete response in only 2-3% of cases. The difference is profound.
KEYNOTE-671 showed similar benefits with perioperative pembrolizumab, achieving 62% event-free survival at two years compared to 40% with chemotherapy alone in stage 2-3B resectable NSCLC.
Who should receive perioperative immunotherapy?
According to current NICE guidance, National Comprehensive Cancer Network recommendations, and guidance from the Canadian Cancer Society, perioperative immunotherapy should be offered to:
-
Stage 2A-3A NSCLC patients fit enough for lung cancer surgery
-
Tumours that are resectable (completely removable) based on staging scans
-
Patients with adequate performance status and overall health to tolerate both treatment and surgery
The typical treatment sequence is:
-
Neoadjuvant phase: 3-4 cycles of platinum-based chemotherapy plus nivolumab or pembrolizumab over 9-12 weeks
-
Surgery: Lobectomy or other lung cancer surgery performed 4-6 weeks after completing neoadjuvant treatment
-
Adjuvant phase: Continuation of immunotherapy alone (no more chemotherapy) for up to one year post-surgery
This approach has now superseded the older paradigm of "surgery first, then decide about adjuvant treatment." For stage 2-3A disease, perioperative immunotherapy is becoming the new standard of care in clinical oncology.
Why does perioperative treatment work better than adjuvant-only?
Several mechanisms likely contribute:
-
Giving immunotherapy before surgery allows the immune system to "learn" about lung cancer cells while the primary lung cancer is still present, potentially creating stronger anti-tumour immunity
-
Neoadjuvant treatment can shrink the tumour, making surgery easier and reducing the risk of positive margins
-
Starting treatment immediately addresses micrometastatic disease (cancer cells that have already spread but aren't visible on scans) earlier
-
Patients tolerate neoadjuvant treatment better than post-surgical adjuvant treatment – completion rates are higher
The implication for patients: if you've been diagnosed with stage 2 or 3A lung cancer and told you need surgery, ask specifically about perioperative immunotherapy. This is no longer experimental – it's evidence-based standard of care that significantly reduces your risk of cancer recurrence and improves lung cancer survival statistics.
Access Advanced Treatment Quickly
SABR treatment within 2 weeks. Biomarker results in 7-14 days. No NHS waiting lists. Start your path to remission now.
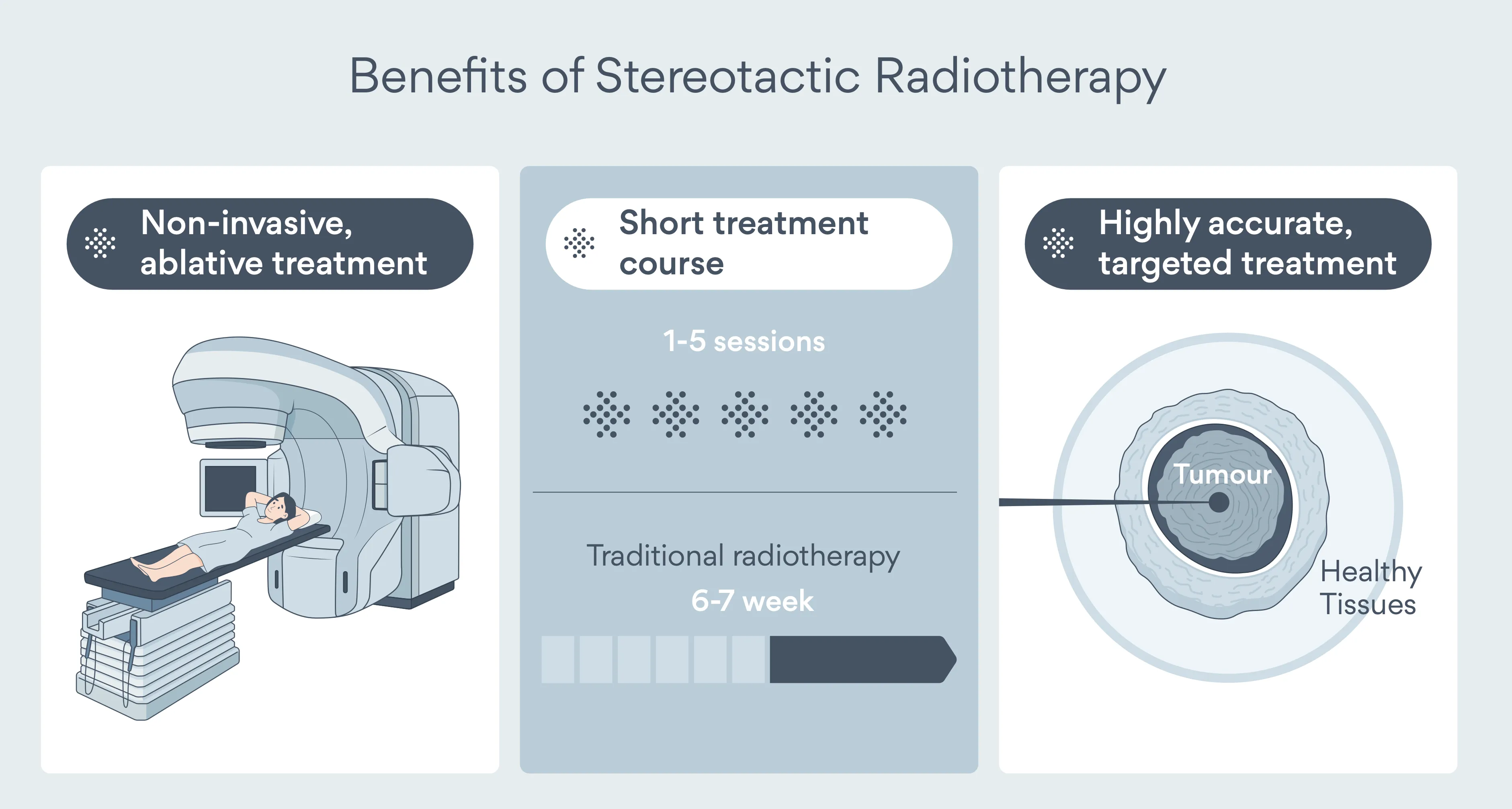
Book Your Same-Day ConsultationStereotactic Radiotherapy (SABR) for Early-Stage Lung Cancer
SABR – also called SBRT (stereotactic body radiation therapy) or stereotactic ablative radiation therapy – delivers very high doses of precisely targeted radiation therapy in just 1-8 sessions, typically over 1-2 weeks.
It's the treatment I offer most frequently in my practice, and the reason is simple: outcomes rival lung cancer surgery, but treatment is non-invasive, completed in days rather than weeks, and suitable for patients who would never tolerate an operation due to lung disease, heart disease, or other conditions affecting overall health.
The data is compelling. The SPACE, CHISEL, and SABRTooth clinical trials (among others) have shown that SABR achieves local control rates of 90-95% at three years for stage 1 NSCLC. In my own practice using CyberKnife, we see 90% local control at five years – meaning lung cancer remission is maintained in 9 out of 10 patients.
What does that mean practically? If you have a 2cm primary lung cancer in the periphery of your lung, and we treat it with SABR, there's a 9 in 10 chance that tumour will never grow back. The small risk of distant recurrence (new tumours developing elsewhere) remains, but the treated tumour itself is eradicated.
Treatment is typically 1-8 sessions:
-
Single-session SABR: For small peripheral tumours in ideal locations, a single 30-minute treatment under the CyberKnife. Completed in one day. Local control >95% at three years.
-
3-session SABR: For slightly larger tumours or those near critical structures. Treatment over one week.
-
5-8 session SABR: For central tumours near major airways or blood vessels, where single-fraction treatment carries higher risk.
Side effects are generally mild – some fatigue, occasional chest wall discomfort if the tumour is very peripheral, and a small risk (5-10%) of radiation pneumonitis (lung inflammation) that usually responds well to steroids if it occurs.
Most lung cancer patients return to normal activities within a week or two.
SABR is increasingly used in stage 4 lung cancer as well, particularly for oligometastatic NSCLC. If you have 1-3 metastases and your systemic therapy has achieved good control, treating those remaining lesions with SABR can convert you from "stage 4 cancer requiring ongoing treatment" to "long-term lung cancer remission with surveillance only."
This approach – aggressive local therapy for all visible disease – is supported by clinical trials like SABR-COMET and NorthStar, and is now incorporated into NHS/NICE guidelines for oligometastatic NSCLC.
Targeted Therapy for Genetic Mutations
We've discussed specific mutations above. The broader strategic point is this: if your cancer has a driver mutation, targeted therapy should be first-line cancer treatment, not chemotherapy.
Sequencing strategy matters. For EGFR-positive disease, osimertinib is now first-line because it overcomes most resistance mechanisms from earlier-generation TKIs. For ALK-positive disease, alectinib or brigatinib are now preferred over crizotinib because they prevent brain metastases and delay resistance.
If first-line targeted therapy eventually stops working, second-line treatment options exist:
-
EGFR: chemotherapy + immunotherapy, or newer TKIs in clinical trials
-
ALK: second-generation TKI → lorlatinib for resistant disease
-
ROS1: chemotherapy or lorlatinib
-
KRAS G12C: chemotherapy + immunotherapy after progression on sotorasib
The goal is sequential therapy – each line of treatment achieving months to years of lung cancer remission, stringing together periods of disease control that extend overall survival well beyond what historical cancer statistics would suggest.
Immunotherapy for Lung Cancer
Checkpoint inhibitors – pembrolizumab, nivolumab, atezolizumab, durvalumab – work by releasing the brakes on your immune system, allowing it to recognise and attack cancerous cells.
When immunotherapy works, it often works durably. The five-year survival statistics for lung cancer patients treated with pembrolizumab show a plateau at around 30% – meaning nearly one in three patients with metastatic NSCLC and high PD-L1 expression remain alive (and mostly in remission) five years after starting treatment.
Some of these lung cancer survivors have stopped immunotherapy and remain in remission off all treatment. This is extraordinarily rare with chemotherapy and represents a complete shift in lung cancer survival outcomes.
When immunotherapy doesn't work, it tends to fail early – progression within the first 3-6 months. This is called primary resistance.
The challenge with immunotherapy is predicting who will respond. PD-L1 expression helps, but it's imperfect. Some patients with PD-L1 <1% respond beautifully; others with PD-L1 ≥90% progress rapidly.
Tumour mutational burden, immune cell infiltration, and other biomarkers add predictive value, but we're not yet at the point where we can reliably say "you will respond" or "you won't."
Combination approaches are increasingly standard:
-
Chemotherapy + pembrolizumab (most common first-line regimen for PD-L1 <50%)
-
Nivolumab + ipilimumab (dual checkpoint blockade, higher toxicity but durable responses)
-
Chemotherapy + immunotherapy for limited-stage small cell lung cancer (modest benefit but now NICE-approved)
Side effects – immune-related adverse events – can affect any organ system: colitis, hepatitis, pneumonitis, thyroid dysfunction, adrenal insufficiency, and (rarely) myocarditis or neurological toxicity. Most are manageable with steroids, but severe cases require permanent discontinuation of immunotherapy.
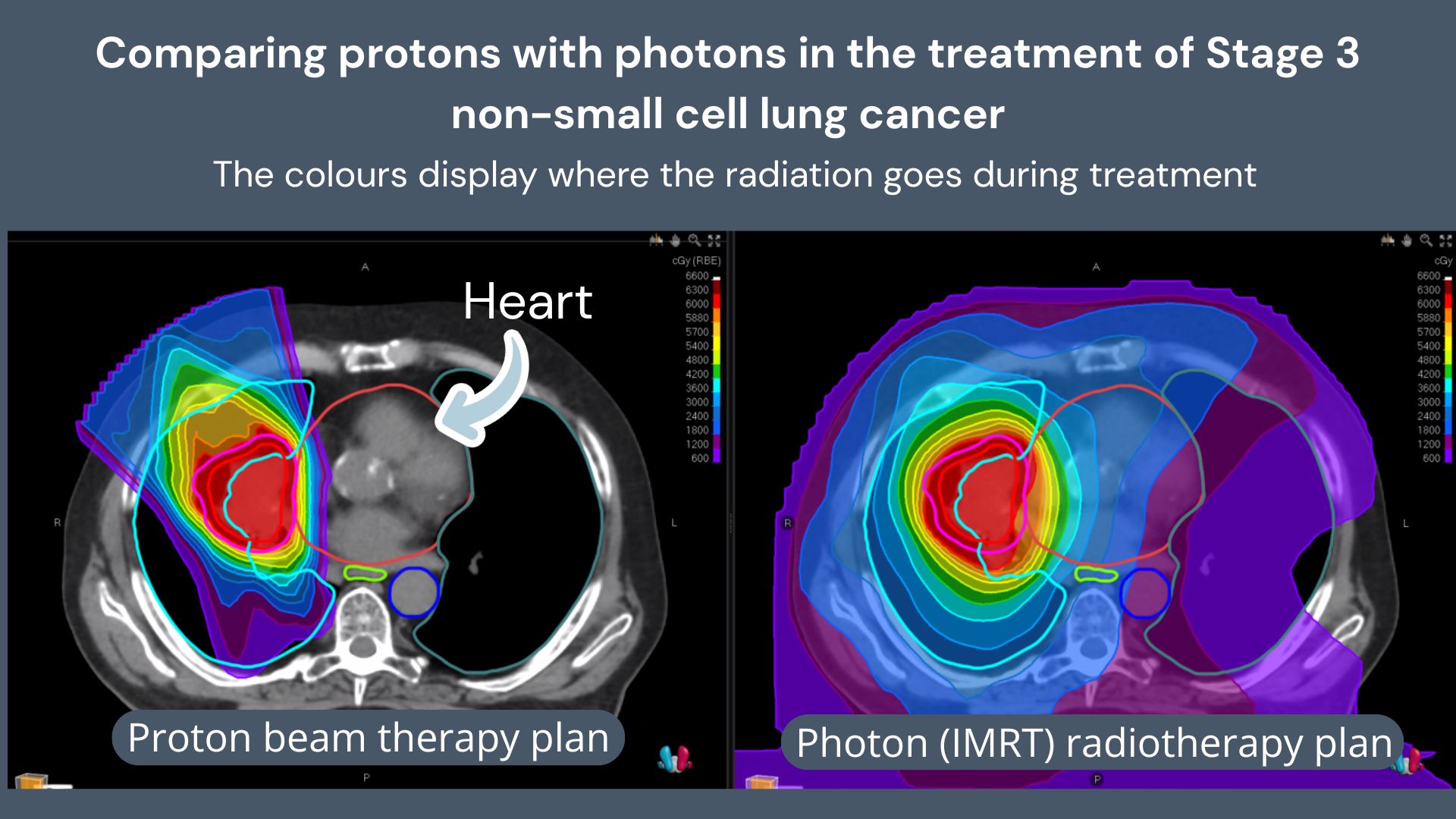
Proton Beam Therapy for Lung Cancer
Proton beam therapy delivers radiation with exceptional precision, depositing almost all its energy at the tumour site and virtually none beyond it. This contrasts with conventional radiation therapy, where X-rays continue through the body, irradiating healthy tissue beyond the target.
For lung cancer, the primary advantage is reduced cardiac and lung toxicity, particularly for centrally located tumours near the heart or major airways – important considerations for patients with existing lung disease or heart conditions that affect overall health.
The UK now has two proton centres: Christie (Manchester), and UCLH (London). Proton therapy is funded by NHS England for selected cases where conventional radiation therapy would deliver excessive dose to critical organs.
Lung cancer remission rates are similar to conventional radiation therapy – local control rates of 85-90% at three years for stage 3 NSCLC treated with concurrent chemoradiotherapy. The benefit is not higher cure rates, but lower side effects, particularly reduced risk of cardiac events in long-term survivors.
Proton therapy is particularly valuable for:
-
Central tumours where conventional SABR would be too risky
-
Re-irradiation (treating a tumour that was previously irradiated)
-
Young lung cancer patients where minimising long-term cardiac risk is paramount
-
Patients with pre-existing heart disease or chronic obstructive pulmonary disease
I work closely with the Proton International London team for patients who would benefit from proton therapy, either as primary treatment or as part of re-irradiation strategies.
Surgery and Adjuvant Therapy
Surgical resection – lobectomy (removing one lobe of the lung) or segmentectomy (removing part of a lobe) – remains the gold standard for early-stage NSCLC in lung cancer patients fit enough to tolerate the operation.
Five-year lung cancer survival rate after lung cancer surgery for stage 1A NSCLC is 77-92%, and most of these patients are cured. They die of other causes decades later, not lung cancer recurrence.
Adjuvant therapy after surgery reduces the risk of cancer recurrence:
-
Adjuvant chemotherapy: Offered for stage 2 and 3 disease (and sometimes high-risk stage 1B). Four cycles of platinum-based chemotherapy reduce recurrence risk by approximately 5-10%.
-
Adjuvant immunotherapy: Atezolizumab for one year after surgery + chemotherapy improves disease-free survival in stage 2-3 NSCLC with PD-L1 ≥1%.
-
Adjuvant targeted therapy: Osimertinib for three years after lung cancer surgery in EGFR-positive stage 1B-3A NSCLC reduces recurrence risk by 80% (ADAURA trial).
The ADAURA results in particular have changed practice fundamentally. If you have early-stage EGFR-positive NSCLC, surgery followed by three years of osimertinib offers an 89% chance of remaining disease-free at three years – outcomes that approach cure in many cases and represent a dramatic improvement in lung cancer remission rates for this molecular subtype.
Treatment for Oligometastatic Lung Cancer
Oligometastatic disease – typically defined as 1-5 metastases that can all be treated with local therapy – represents a unique opportunity for aggressive cancer treatment with curative intent.
The treatment paradigm is:
-
Systemic therapy first (chemotherapy, targeted therapy, or immunotherapy) to control microscopic disease and assess cancer biology
-
Local therapy to all visible metastases (SABR, surgery, or other ablative techniques) if systemic disease remains controlled
-
Ongoing systemic therapy or surveillance depending on response and mutation status
In the SABR-COMET-10 clinical trial, patients with 1-3 metastases from any cancer type (including lung) who received SABR to all sites had a median survival of 50 months compared to 28 months with systemic therapy alone – a remarkable improvement in overall survival.
The NorthStar trial specifically demonstrated this benefit for EGFR-positive metastatic NSCLC, showing median PFS of 25.3 months with osimertinib plus local consolidative therapy versus 17.5 months with osimertinib alone.
We now know that some lung cancer patients with oligometastatic NSCLC – particularly those with EGFR or ALK mutations, or high PD-L1 expression – can achieve lung cancer remission lasting 3-5 years or longer with this approach.
Practical example: You have stage 4 lung adenocarcinoma with an EGFR mutation. Your primary lung cancer is in the right upper lobe, and you have two brain metastases and one adrenal metastasis. You start osimertinib. After three months, all tumours have shrunk by 70%. We now have several treatment options:
-
Continue osimertinib indefinitely (standard approach)
-
Treat all four lesions with SABR while continuing osimertinib (oligometastatic approach)
-
Treat brain metastases with stereotactic radiosurgery, continue osimertinib (hybrid approach)
The oligometastatic approach – SABR to all four sites while continuing osimertinib – may extend your lung cancer remission from 12-18 months (typical with osimertinib alone) to 25-40 months based on the NorthStar data, fundamentally changing your lung cancer survival rate.
This is not suitable for everyone. It requires good performance status and overall health, limited sites of disease, and cancer that responds well to systemic therapy. But when appropriate, it can be transformative.
Explore Oligometastatic Treatment
If you have 1-5 metastases, aggressive local treatment combined with systemic therapy may extend your remission. Let's assess if you're a candidate.
Discuss Your Oligometastatic OptionsHow Long Does Lung Cancer Remission Last?
This is the question lung cancer patients ask most often, and the answer is frustratingly variable: anywhere from months to decades, depending on the factors below.
Factors That Influence Remission Duration
Cancer biology and growth rate
Some lung cancers are indolent – they grow slowly and respond well to treatment. Others are aggressive, developing resistance quickly and progressing despite multiple therapies.
Histological subtype matters: lepidic-predominant lung adenocarcinoma (the slowest-growing NSCLC subtype) can remain in remission for years even without treatment. Small cell lung cancer, conversely, almost always experiences recurrence despite aggressive initial therapy.
Genetic mutations influence durability. EGFR and ALK-positive cancers respond dramatically to targeted therapy but often develop resistance within 1-3 years. Immunotherapy-responsive cancers may remain in remission for 5+ years, sometimes indefinitely.
Treatment type and response
Surgery or SABR for stage 1 NSCLC typically results in permanent remission – functional cure. If cancer recurrence occurs, it's usually a second cancer (new primary tumour) or distant metastasis, not regrowth of the treated lesion.
Targeted therapy for stage 4 EGFR-positive NSCLC achieves a median progression-free survival of 17-19 months on osimertinib alone, though the NorthStar trial showed this can extend to 25-40 months when combined with local consolidative therapy. This means half of lung cancer patients remain in remission longer than that, some for 3-5 years.
Immunotherapy is less predictable. Some lung cancer patients progress within months (primary resistance). Others achieve durable lung cancer remission lasting 5+ years.
The depth of response matters. Patients who achieve complete remission (no evidence of disease) stay in remission longer than those with partial remission (significant shrinkage but residual disease visible).
Genetic mutations
We've covered this extensively above, but it bears repeating: EGFR, ALK, ROS1, and other driver mutations predict both initial response to targeted therapy and eventual resistance.
First-line osimertinib for EGFR-positive disease: median remission ~18 months, but with the addition of local consolidative therapy this extends to 25-40 months. Some lung cancer patients remain progression-free for 4-5 years.
First-line alectinib for ALK-positive disease: median remission ~35 months, with a subset remaining progression-free beyond five years.
PD-L1 high expression + immunotherapy: median remission ~8-10 months, but the tail of the survival curve shows 25-30% remaining progression-free at five years.
Overall health and lifestyle
Lung cancer patients with good performance status (able to care for themselves, active most of the day) remain in remission longer than those with poor performance status.
Smoking cessation extends remission duration and reduces lung cancer risk factors for second cancers. Former smokers who quit at diagnosis have better outcomes than those who continue smoking.
Comorbidities matter. Patients with significant heart disease, chronic obstructive pulmonary disease, or diabetes have shorter remissions, partly because they tolerate cancer treatment less well and partly because systemic inflammation may fuel cancer progression.
Median Remission Duration by Treatment Type
| Treatment | Cancer Type | Median Remission Duration | 5-Year Remission Rate |
|---|---|---|---|
| Surgery | Stage 1 NSCLC | Permanent (functional cure) | 70-85% |
| SABR | Stage 1 NSCLC | Permanent (functional cure) | 75-90% |
| Perioperative nivolumab + surgery | Stage 2-3A NSCLC | Not yet mature | ~74% event-free survival at 3 years |
| Adjuvant osimertinib (3 years) | EGFR+ stage 1B-3A NSCLC post-surgery | Not yet mature | ~89% disease-free survival at 3 years |
| Osimertinib alone | EGFR+ metastatic NSCLC | 17.5 months | 15-20% |
| Osimertinib + local consolidative therapy | EGFR+ metastatic NSCLC | 25.3 months (up to 39.8 for ex19del) | Data pending |
| Alectinib | ALK+ metastatic NSCLC | 34.8 months | 40-50% |
| Pembrolizumab | PD-L1 ≥50% metastatic NSCLC | 10.3 months | 25-32% |
| Chemotherapy + immunotherapy | PD-L1 <50% metastatic NSCLC | 6-8 months | 10-15% |
| Concurrent chemoradiotherapy + durvalumab | Stage 3 NSCLC | 17-28 months | 36-43% |
| Chemotherapy + radiation therapy | Limited-stage small cell lung cancer | 12-18 months | 20-30% |
These figures represent median values – half of lung cancer patients do better, half do worse. Individual outcomes depend on the factors discussed above.
What Happens When Lung Cancer Remission Ends?
Most patients with stage 4 lung cancer eventually experience progression – new tumours appearing, or existing tumours beginning to grow despite treatment. This doesn't mean the end of treatment choices. Modern lung cancer therapy is sequential: when one approach stops working, we move to the next.
Second-Line Treatment Options
For EGFR-positive NSCLC after osimertinib progression:
-
Chemotherapy (platinum doublet + immunotherapy if not previously used)
-
Clinical trials of next-generation EGFR inhibitors
-
Local therapy (SABR) for oligoprogression (1-3 sites of progression while other metastases remain controlled)
For ALK-positive NSCLC after alectinib progression:
-
Lorlatinib (third-generation ALK inhibitor, highly effective for brain metastases)
-
Chemotherapy + immunotherapy
-
Lung cancer research clinical trials
For immunotherapy-refractory NSCLC:
-
Chemotherapy (docetaxel or pemetrexed, depending on histology)
-
Clinical trials of novel agents (bispecific antibodies, antibody-drug conjugates, etc.)
For small cell lung cancer after first-line progression:
-
Topotecan chemotherapy
-
Lurbinectedin (recently approved in UK)
-
Clinical trials
The principle throughout is to maintain quality of life while controlling disease. Some lung cancer patients receive 3-4 lines of systemic therapy, stringing together periods of remission that extend overall survival to 3-5 years in stage 4 disease.
Re-Treatment with SABR for New Lung Nodules
If you develop a new lung nodule years after successful treatment of your initial primary lung cancer, SABR can often treat it as effectively as the first time.
This is common in patients with smoking-related lung damage – the entire lung has been exposed to carcinogens, so second cancers can develop even after the first cancer was cured.
In my practice, I've treated lung cancer patients with 2-3 separate lung cancers over a span of 5-10 years, each treated with SABR, each achieving long-term control. These lung cancer survivors remain alive and well, despite having had multiple lung cancers.
SABR can also treat oligoprogression – when 1-2 metastases begin growing while everything else remains controlled on systemic therapy. Treating those progressive lesions with SABR while continuing the same systemic therapy can extend lung cancer remission by months to years.
Clinical Trials for Resistant Lung Cancer
When standard therapies have been exhausted, clinical trials offer access to experimental treatments that may become standard of care in the future.
The UK has an active lung cancer trial landscape through translational lung cancer research programmes coordinated by the National Cancer Research Institute (NCRI) Lung Cancer Group and commercial clinical trials run by pharmaceutical companies.
Current areas of investigation include:
-
Next-generation targeted therapies for resistant EGFR, ALK, and KRAS mutations
-
Antibody-drug conjugates that deliver chemotherapy directly to lung cancer cells
-
Bispecific antibodies that link cancer cells to immune cells
-
CAR-T cell therapy for solid tumours (early stages)
-
Combination immunotherapy strategies to overcome resistance
I maintain close relationships with the thoracic oncology teams at UCL, Royal Marsden, and Guy's & St Thomas's, and can facilitate rapid clinical trial referrals when appropriate.
Palliative Care to Maintain Quality of Life
Palliative care has nothing to do with "giving up." It's the medical specialty focused on symptom control, quality of life, and supporting lung cancer patients through serious illness.
Modern palliative care is given alongside active cancer treatment, not instead of it. Cancer patients who receive early palliative care involvement have better quality of life, fewer emergency admissions, and (in several studies) live longer than those who receive oncology care alone.
Palliative care teams can help with:
-
Pain management
-
Breathlessness
-
Fatigue
-
Nausea and appetite loss
-
Anxiety and depression
-
Advanced care planning
-
Family support
If you're receiving active lung cancer treatment, particularly for advanced lung cancer, asking for palliative care input is not a sign of decline – it's a way of ensuring you feel as well as possible throughout treatment.
Monitoring Your Lung Cancer for Recurrence
After achieving lung cancer remission, lifelong surveillance is essential. The goal is to detect lung cancer recurrence early, when it's most treatable. Lung cancer screening programmes are also increasingly used to detect new primary cancers in high-risk individuals, even after successful treatment of an initial lung cancer.
Standard Monitoring: CT Scans and Clinical Exams
NHS standard surveillance:
-
CT chest every 3-4 months for the first 2 years
-
Every 6 months for years 3-5
-
Annually thereafter
Private practice surveillance (my typical approach):
-
CT chest + upper abdomen every 3 months for the first year
-
Every 3-4 months for year 2
-
Every 4-6 months for years 3-5
-
Every 6-12 months thereafter
Lung cancer patients with EGFR or ALK-positive disease receiving adjuvant targeted therapy are monitored more frequently – typically every 3 months while on treatment.
Brain MRI is added to surveillance protocols for ALK-positive disease (high risk of CNS metastases) and for patients with stage 3 disease treated with concurrent chemoradiotherapy.
Advanced Blood Monitoring: Circulating Tumour DNA (ctDNA)
Circulating tumour DNA (ctDNA) testing detects tiny fragments of cancer DNA in your bloodstream. It's far more sensitive than CT imaging – it can detect cancer recurrence months before a tumour becomes visible on a scan.
This is transformative for lung cancer patients in remission. The psychological burden of "scanxiety" – the fear and anxiety before each surveillance scan – is significant. Blood tests every 6-8 weeks between scans provide reassurance that cancer remains undetectable.
If ctDNA becomes positive, it signals lung cancer recurrence before scans show anything. This provides a window of opportunity to intensify surveillance, repeat imaging with PET-CT, or (in some cases) restart treatment preemptively.
ctDNA monitoring offers several advantages:
Earlier detection of recurrence: Studies show ctDNA becomes detectable an average of 3-6 months before recurrence appears on CT scans. This lead time allows for earlier intervention.
Treatment response monitoring: For patients on targeted therapy or immunotherapy, ctDNA levels can indicate whether treatment is working before the next scheduled scan. Rising ctDNA levels suggest resistance is developing; falling levels confirm response.
Personalised surveillance: Patients with persistently negative ctDNA may be able to extend intervals between CT scans, reducing radiation exposure and cost. Those with detectable ctDNA warrant more intensive monitoring.
Mutation profiling: ctDNA testing can identify resistance mutations (like EGFR T790M or C797S) that emerge during targeted therapy, guiding selection of next-line treatment.
I offer ctDNA monitoring as part of comprehensive surveillance for lung cancer patients, typically testing every 2-3 months during the first 2 years of remission when recurrence risk is highest.
The cost varies per test depending on the panel used. For patients with EGFR, ALK, or other targetable mutations, the investment often provides valuable peace of mind and earlier detection of resistance. You can learn more, including costs, on my cancer blood tests page.
This isn't for every cancer patient – some patients find the anxiety of a potentially positive blood test counterproductive, or prefer the certainty of imaging alone. But for those who want maximum reassurance between scans, or who have high-risk features (stage 3 disease, close surgical margins, positive lymph nodes), ctDNA monitoring offers meaningful clinical value.
Get Comprehensive Surveillance
CT surveillance every 3 months plus ctDNA blood monitoring for earliest detection. Results between scans to minimise scanxiety.
Enhance Your Follow-Up CareSymptoms That Require Urgent Review
Between scheduled scans, contact your oncologist immediately if you develop lung cancer symptoms including:
-
New persistent cough or worsening of existing cough
-
Haemoptysis (coughing up blood, even small amounts)
-
New or worsening breathlessness not explained by other causes
-
Unexplained weight loss (>5% body weight over 3 months)
-
New bone pain, particularly back pain or pain at night
-
Persistent headaches with nausea, vision changes, or personality changes
-
New neurological symptoms (weakness, numbness, speech difficulty, balance problems)
-
Jaundice (yellowing of skin/eyes) or severe fatigue suggesting liver involvement
Most of these symptoms turn out to be unrelated to cancer. But in someone with a history of lung cancer, urgent investigation is warranted to rule out recurrent lung cancer.
Don't wait for your next scheduled scan. An urgent CT or PET-CT can be arranged within 24-48 hours if there's clinical concern.
Living Well During Lung Cancer Remission
Remission is not the same as being cancer-free. The psychological adjustment can be surprisingly difficult – many lung cancer survivors find the transition from active treatment to surveillance more challenging than treatment itself.
Managing Physical Side Effects After Treatment
Fatigue is the most common persistent symptom after lung cancer treatment. It typically improves over 6-12 months but may never fully resolve.
Graded exercise – starting with short walks and gradually increasing duration and intensity – is the most effective intervention. Physiotherapy and pulmonary rehabilitation programmes can help rebuild fitness.
Breathlessness after lung cancer surgery or radiation therapy reflects reduced lung capacity. Pulmonary function typically stabilises 3-6 months after treatment. Breathing exercises, pursed-lip breathing, and optimising body position can all help manage dyspnoea.
If you develop progressive breathlessness months or years after radiation therapy, radiation pneumonitis or pulmonary fibrosis may be responsible. This usually responds to steroids, though some scarring may be permanent.
Radiation pneumonitis (lung inflammation) occurs in 5-15% of patients after SABR or conventional radiation therapy. Symptoms include dry cough, breathlessness, and fatigue, typically 2-6 months after treatment. Most cases are mild and resolve spontaneously. Moderate cases respond well to prednisolone. Severe cases require hospitalisation and extended steroid courses.
Peripheral neuropathy (numbness and tingling in hands and feet) is common after platinum-based chemotherapy. It peaks at 3-6 months after treatment and usually improves slowly over 1-2 years, though some patients have permanent residual symptoms.
Emotional Challenges of Remission
Scanxiety – the fear and anxiety before each surveillance scan – affects most lung cancer patients. It typically intensifies 1-2 weeks before the scan and resolves once results are discussed.
Strategies that help:
-
Scheduling scans first thing in the morning so you're not waiting all day
-
Arranging results appointments within 48-72 hours of the scan (my standard practice)
-
ctDNA blood monitoring between scans for additional reassurance
-
Mindfulness, meditation, or formal CBT for health anxiety
Fear of cancer recurrence is universal and doesn't disappear with time. Some lung cancer survivors find that anxiety lessens after the 2-year mark (when recurrence risk drops substantially), but others remain hypervigilant for years.
Support groups – either in-person through Roy Castle Lung Cancer Foundation or online through forums and Facebook groups – can be invaluable. Speaking with others who understand the experience is often more therapeutic than speaking with clinicians. Many lung cancer survivors find a free lung cancer guide from charities like Cancer Research UK or the American Lung Association helpful for understanding their journey.
If fear of recurrence is interfering with daily life – causing avoidance behaviours, sleep disturbance, or relationship strain – formal psychological support through a cancer centre's psychology service or a private psycho-oncology specialist is warranted.
Survivor guilt affects some lung cancer survivors, particularly those in peer support groups where others have experienced cancer recurrence or died. This is normal and doesn't mean you're ungrateful for your remission. Processing these feelings with a therapist or counsellor can help.
Lifestyle Modifications to Support Remission
Smoking cessation is non-negotiable. If you're still smoking, stopping now – even after diagnosis – extends survival, improves lung cancer treatment outcomes, and reduces lung cancer risk factors for second cancers.
The NHS Stop Smoking Service provides free support, including nicotine replacement therapy and varenicline (Champix). Vaping is an acceptable harm-reduction strategy if it helps you stop smoking combustible tobacco.
Nutrition doesn't need to be complicated. A Mediterranean-style diet – vegetables, fruits, whole grains, oily fish, olive oil – is associated with better cancer outcomes in multiple studies.
Avoid excessive red or processed meat. Limit alcohol. Maintain a healthy weight if possible, though weight loss during or immediately after cancer treatment is common and doesn't need to be aggressively corrected if you're otherwise stable and maintaining overall health.
There's no evidence that specific "anti-cancer" diets (ketogenic, alkaline, juice cleanses, etc.) improve lung cancer outcomes. Some are actively harmful, causing malnutrition during a period when your body needs adequate protein and calories to recover.
Exercise is the single most evidence-based lifestyle intervention for cancer survivors. Regular physical activity reduces fatigue, improves mood, strengthens immune function, and (in several studies) reduces cancer recurrence risk.
Aim for 150 minutes of moderate aerobic activity per week – brisk walking, cycling, swimming. Resistance training twice a week helps rebuild muscle mass lost during treatment.
If you're deconditioned after cancer treatment, start slowly. A 10-minute walk daily is better than nothing. Pulmonary rehabilitation programmes are available through the NHS and can provide structured, supervised exercise tailored to your fitness level.
Stress management matters, though the evidence linking stress directly to cancer outcomes is weaker than many lung cancer patients believe. Nonetheless, chronic stress impairs immune function and quality of life.
Practices that help: mindfulness meditation, yoga, tai chi, spending time in nature, maintaining social connections, pursuing hobbies and interests outside cancer.
If you're experiencing clinical anxiety or depression – persistent low mood, anhedonia, sleep disturbance, suicidal thoughts – speak to your GP. Antidepressants and CBT are both effective and don't interfere with cancer treatment.
Does Smoking History Affect Lung Cancer Remission Rates?
Yes, but not as dramatically as many lung cancer patients fear.
Never-smokers with lung cancer tend to have better outcomes than current or former smokers, for several reasons:
-
Their cancers are more likely to carry EGFR or ALK mutations (which respond well to targeted therapy)
-
They have better baseline lung function, tolerating cancer treatment more easily
-
They're less likely to have comorbidities like chronic obstructive pulmonary disease or cardiovascular disease that affect overall health
In the FLAURA trial, median progression-free survival on osimertinib was 21 months in never-smokers compared to 17 months in former smokers.
Former smokers who quit at diagnosis have outcomes similar to never-smokers in many studies. The lung's repair mechanisms begin immediately after stopping smoking. Inflammation decreases, immune function improves, and treatment tolerance increases.
Even lung cancer patients who've smoked heavily for decades see meaningful benefit from stopping at diagnosis.
Current smokers have worse outcomes, primarily because:
-
Smoking impairs wound healing after lung cancer surgery
-
Smoking increases radiation therapy toxicity (higher risk of pneumonitis)
-
Smoking reduces chemotherapy effectiveness
-
Smoking increases cardiovascular risk, making some treatments more dangerous
-
Continued smoking increases lung cancer risk factors for second cancers
Stopping smoking at any point – even after diagnosis – improves outcomes. It's never too late.
Frequently Asked Questions About Lung Cancer Remission
What is the difference between remission and cure in lung cancer?
Remission means your cancer has responded to cancer treatment and is no longer detectable (complete remission) or has shrunk significantly (partial remission). You remain under surveillance with regular scans because cancer recurrence is possible.
Cure means the cancer will never come back. We typically use this term for lung cancer patients who remain disease-free 5+ years after treatment for stage 1-2 disease. Beyond five years, recurrence risk is very low (though not zero).
The challenge is that we can't definitively say someone is cured until many years have passed. A lung cancer patient in complete remission for three years has an excellent prognosis, but we wouldn't yet call them cured.
For practical purposes: if you've had lung cancer surgery or SABR for stage 1 NSCLC and remain clear on scans for five years, you're functionally cured. Your chance of dying from lung cancer is now very low – you're much more likely to die of heart disease, stroke, or a second cancer decades later.
Can stage 4 lung cancer go into remission?
Yes, absolutely. Complete remission is less common in stage 4 lung cancer than in early-stage cancer, but partial remission – tumours shrinking by 50-80% and remaining stable – is routine with modern cancer treatment.
For lung cancer patients with EGFR or ALK mutations, targeted therapy achieves objective response in 70-80% of cases. Some remain in lung cancer remission for 3-5 years or longer, particularly when local consolidative therapy is added to all sites of disease.
For patients with high PD-L1 expression (≥50%), immunotherapy achieves complete remission in 10-15% and partial remission in another 30-35%.
The key distinction: remission in stage 4 lung cancer usually requires ongoing treatment. You don't finish chemotherapy and stop – you continue targeted therapy or immunotherapy indefinitely, or until progression. Some call this "disease control" rather than remission.
However, a subset of lung cancer patients with oligometastatic disease (1-5 metastases) treated aggressively with systemic therapy + SABR to all sites achieve durable remission without ongoing treatment. The NorthStar trial demonstrated this specifically for EGFR-positive disease, with comprehensive local consolidative therapy extending median PFS to 33 months. This is now an established treatment paradigm and is covered in detail on my stage 4 cancer remission page.
What are the early signs of lung cancer remission?
Clinical signs that cancer treatment is working:
-
Improved energy and appetite within 2-4 weeks of starting treatment
-
Resolution of lung cancer symptoms (cough, breathlessness, pain)
-
Stable or increasing weight after initial treatment-related loss
-
Blood tumour markers (if elevated at diagnosis) trending downward
Radiological response is usually assessed after 6-12 weeks of systemic therapy:
-
Complete response: no visible tumour on CT or PET scan
-
Partial response: tumour shrinks by ≥30%
-
Stable disease: tumour neither grows nor shrinks significantly
Most oncologists use PET-CT to assess response after concurrent chemoradiotherapy for stage 3 disease, and CT alone for monitoring targeted therapy or immunotherapy in stage 4 lung cancer.
For SABR-treated early-stage lung cancer, the first surveillance scan at 3 months often shows inflammation around the treated area, making it difficult to assess response. By 6-12 months, this inflammation resolves, and you can see clear evidence of tumour control (shrinkage or scar tissue replacing the original tumour).
Is lung cancer remission possible without chemotherapy?
Yes, and increasingly common for lung cancer patients.
For early-stage disease:
-
Lung cancer surgery alone achieves cure in many stage 1 patients
-
SABR alone achieves equivalent outcomes without chemotherapy
For EGFR, ALK, ROS1-positive stage 4 lung cancer:
-
Targeted therapy alone (osimertinib, alectinib, etc.) is first-line cancer treatment
-
Chemotherapy is reserved for progression after targeted therapy
For PD-L1 high (≥50%) metastatic NSCLC:
-
Pembrolizumab monotherapy achieves lung cancer remission in 40-45% without chemotherapy
-
Chemotherapy is added if immunotherapy alone is insufficient
The paradigm has shifted: chemotherapy is no longer automatically first-line treatment for lung cancer. Biomarker testing guides personalised therapy, and many lung cancer patients never need chemotherapy, or delay it for months to years.
How often will I need scans during lung cancer remission?
NHS standard:
-
Every 3-4 months for years 1-2
-
Every 6 months for years 3-5
-
Annually thereafter
My typical private practice protocol:
-
Every 3 months for year 1
-
Every 3-4 months for year 2
-
Every 4-6 months for years 3-5
-
Every 6-12 months thereafter, potentially indefinitely
Lung cancer patients on adjuvant targeted therapy (e.g. osimertinib after surgery for EGFR-positive disease) are scanned every 3 months while on treatment.
Patients with oligometastatic disease treated with SABR are monitored more intensively – typically every 2-3 months for the first 2 years.
ctDNA blood monitoring every 2-3 months between scans provides additional surveillance without radiation exposure.
Can lung cancer come back after 5 years of remission?
Yes, though it's uncommon.
Late recurrence (>5 years after initial treatment) occurs in approximately 5-10% of lung cancer patients with early-stage NSCLC. Some of these are true recurrent cancer (the original cancer returning), while others are second cancers – new primary lung cancers (particularly in patients with smoking history and ongoing lung cancer risk factors).
The risk varies by stage:
-
Stage 1: ~5-10% late recurrence rate
-
Stage 2: ~10-15% late recurrence rate
-
Stage 3: ~15-30% late recurrence rate
For stage 4 lung cancer, most recurrences occur within 2-3 years. Lung cancer patients who remain in remission beyond five years are unusual and likely cured (or living with very indolent disease that may never progress).
This is why lifelong surveillance is recommended, even for early-stage lung cancer survivors who were ostensibly cured. Annual CT scans after year 5 allow detection of late recurrence or second cancers when they're still treatable. For high-risk patients with significant smoking history, participation in lung cancer screening programmes may continue indefinitely.
Take Control of Your Treatment Today
Same or next-day appointments. Clear answers within 48 hours. Treatment started ASAP. Call 020 7993 6716 or complete our contact form here.
Book Your Consultation NowDr James Wilson's Approach to Lung Cancer Remission
Rapid Access to Advanced Lung Cancer Treatment
One of the greatest frustrations lung cancer patients express when contacting my practice is the time they've already lost waiting for NHS appointments, waiting for scans, waiting for MDT discussions, waiting for treatment to begin.
In lung cancer, time matters. Fast-growing tumours can progress from stage 1 to stage 3 in a matter of months. Treatment delays allow cancer to spread and can negatively impact lung cancer remission rates.
My practice is built around eliminating those delays:
-
Same-day or next-day consultations for new referrals
-
Scans arranged within 48-72 hours (CT, PET-CT, brain MRI)
-
Biomarker testing results within 7-10 days (EGFR, ALK, ROS1, PD-L1, comprehensive genomic profiling)
-
Treatment planning completed within a week of receiving scan and biopsy results
-
SABR treatment delivered within 2-3 weeks of first consultation
For lung cancer patients whose NHS pathway has been derailed by waiting times, private consultation allows rapid clarification of diagnosis, staging, and treatment options. You can then either proceed with private cancer treatment or return to the NHS with a clear plan and all investigations already completed.
Many patients use private consultation for speed but receive actual treatment on the NHS once the urgency of waiting lists has been bypassed.
SABR Expertise: 90% Control Rate in Early-Stage Lung Cancer
I've treated hundreds of lung cancer patients with early-stage disease using CyberKnife SABR, achieving local control rates of 90% at five years – outcomes equivalent to lung cancer surgery but without the recovery time, lung function loss, or surgical risks.
Most of my SABR patients are treated in 1-3 sessions over 1-2 weeks and return to normal activities immediately.
For oligometastatic disease, I've successfully treated lung cancer patients with up to five separate lung metastases using SABR, often in combination with systemic therapy, achieving durable lung cancer remission in carefully selected cases. The NorthStar trial has now validated this approach specifically for EGFR-positive disease, demonstrating median PFS of 25.3 months with comprehensive local consolidative therapy – outcomes I've been seeing in my practice for several years.
This is particularly relevant for patients whose lung cancer has progressed on first-line systemic therapy but remains limited to 1-3 sites. Aggressive local therapy with SABR while switching to second-line systemic treatment can extend remission by months to years and improve overall survival.
Coordinated Care Across London's Leading Cancer Hospitals
I hold practising privileges at:
-
The Cromwell Hospital (SABR, consultation, chemotherapy)
-
Proton International London (proton beam therapy, re-irradiation)
-
The London Clinic (consultation, chemotherapy)
This allows me to match cancer treatment to your specific needs. CyberKnife SABR is delivered at the Cromwell or (for selected cases) at dedicated SABR centres like Genesis Care. Proton therapy for central tumours or re-irradiation is delivered at Sevenoaks. Chemotherapy and immunotherapy infusions can be arranged at any of these centres, whichever is most convenient for you.
Systemic therapy prescribing, blood monitoring, and symptom management are coordinated through my practice, ensuring continuity even when treatment is delivered across multiple sites.
Personalised Treatment Based on Your Cancer's Genetic Profile
Every lung cancer patient with newly diagnosed NSCLC should have comprehensive biomarker testing before starting treatment. This includes at minimum:
-
EGFR mutation analysis
-
ALK rearrangement testing
-
ROS1 rearrangement testing
-
PD-L1 expression (immunohistochemistry)
For patients with lung adenocarcinoma and no smoking history, or those whose cancer is resistant to standard therapy, I recommend comprehensive genomic profiling with panels like Foundation One CDx or Guardant360, which test for:
-
KRAS (including G12C), BRAF, MET exon 14 skipping, RET, NTRK, HER2
-
Tumour mutational burden (TMB)
-
Microsatellite instability (MSI)
This costs £2,000-3,000 but can identify actionable mutations in 15-20% of lung cancer patients who would otherwise receive empiric chemotherapy.
If you have stage 4 lung cancer, finding a targetable mutation changes everything – remission duration, quality of life during treatment, and overall survival can all improve dramatically with matched targeted therapy. Your lung cancer remission rate may be substantially higher than general population cancer statistics suggest.
Take the Next Step Toward Lung Cancer Remission
If you've been diagnosed with lung cancer, or if you're experiencing lung cancer symptoms and concerned about the possibility, I can see you for a comprehensive consultation within 24-48 hours.
During your first appointment, we'll:
-
Review your imaging, pathology, and any previous treatments in detail
-
Discuss your diagnosis, stage, and prognosis clearly and honestly
-
Arrange any additional investigations needed (biomarker testing, PET-CT, brain MRI)
-
Develop a personalised treatment plan based on your cancer's biology and your priorities
-
Provide written cancer information you can take away and discuss with family
If SABR is appropriate, I'll explain the procedure, expected outcomes, and timeline for cancer treatment. If systemic therapy is needed first, I'll coordinate that and arrange SABR for any residual or oligometastatic disease once response has been assessed.
If your lung cancer has progressed despite previous treatment, we'll discuss second-line and third-line treatment choices, clinical trial opportunities, and strategies to maintain quality of life.
To arrange a consultation:
-
Call my practice directly: 020 79936716
-
Email info@drjameswilson.co.uk
-
Complete the contact form on my website
Consultations are available at The Cromwell Hospital (Kensington), The London Clinic (Marylebone), or via secure video call if distance makes in-person attendance difficult initially.
I respond to all enquiries within 24 hours, typically sooner.
Standard consultation fee: £450
SABR treatment costs vary depending on complexity (single vs multi-fraction) and are discussed during your consultation. Cancer treatment can often be covered by private medical insurance (BUPA, AXA PPP, Aviva, etc.).
Lung cancer is frightening. The NHS waiting times make it worse. My practice exists to give you rapid access to clinical oncology expertise, clear cancer information, and treatment that maximises your lung cancer remission rate.
Let's start that conversation today.
Start Your Journey to Remission
Consultations available at The Cromwell Hospital, The London Clinic, or via secure video call. Respond to all enquiries within 24 hours. £450 consultation fee.
Arrange Your Consultation – 020 7993 6716
Book now